Stuck-in-the-Middle Venturing Strategies Can Hurt You and Your Customers
Simon Hensellek of the Technical University of Dortmund discusses “Beneficial, Harmful, or Both? Effects of Corporate Venture Capital and Alliance Activity on Product Recalls,” which he, David Bendig, and Julian Schulte published in Entrepreneurship, Theory and Practice.
Can established firms successfully source innovation capital from external partners?
When looking at current numbers of external corporate venturing activities, one would supposedly say yes: Corporate venture capital (CVC) investments showed a more than tenfold increase over the past decade up to a record high of $169.3 billion in 2021. Similarly, 56 percent of CEOs in the annual KPMG CEO survey stated that strategic alliances are a fundamental part of their innovation strategy.
However, we also saw other numbers increasing to all-time highs in recent years: the number of failed products that were recalled from the market. In the US alone, the number of drug recalls reached an all-time high of 2,163 in 2019. This is of ample public importance because these products were potentially harmful for consumers’ health. Moreover, recalls can also severely negatively impact the recalling firm. A prominent recall example is Merck’s arthritis drug Vioxx. Taken by over four million Americans, Vioxx caused cardiovascular problems for many, leading to an estimated 140,000 heart attacks and 60,000 deaths. Merck’s market capitalization dropped $25 billion (nearly 27 percent) on the day the recall was announced.

In our study, we combined both observations through a resource-based learning perspective. Since knowledge and innovation sourcing via external venturing is a highly complex endeavor with many pitfalls, our idea was that external venturing might have downsides for the involved firms in the form of more product failure and thus a higher recall likelihood.
By considering firms’ CVC and alliance activities as potential root causes for product recalls, our study is among the first to investigate the relationship between external venturing in a more nuanced way beyond mere financial performance effects. Indeed, we believe that (drug) recalls might be one of the most meaningful dependent variables considered in the field of corporate venturing because they not only hurt the recalling firm but can harm the general public.
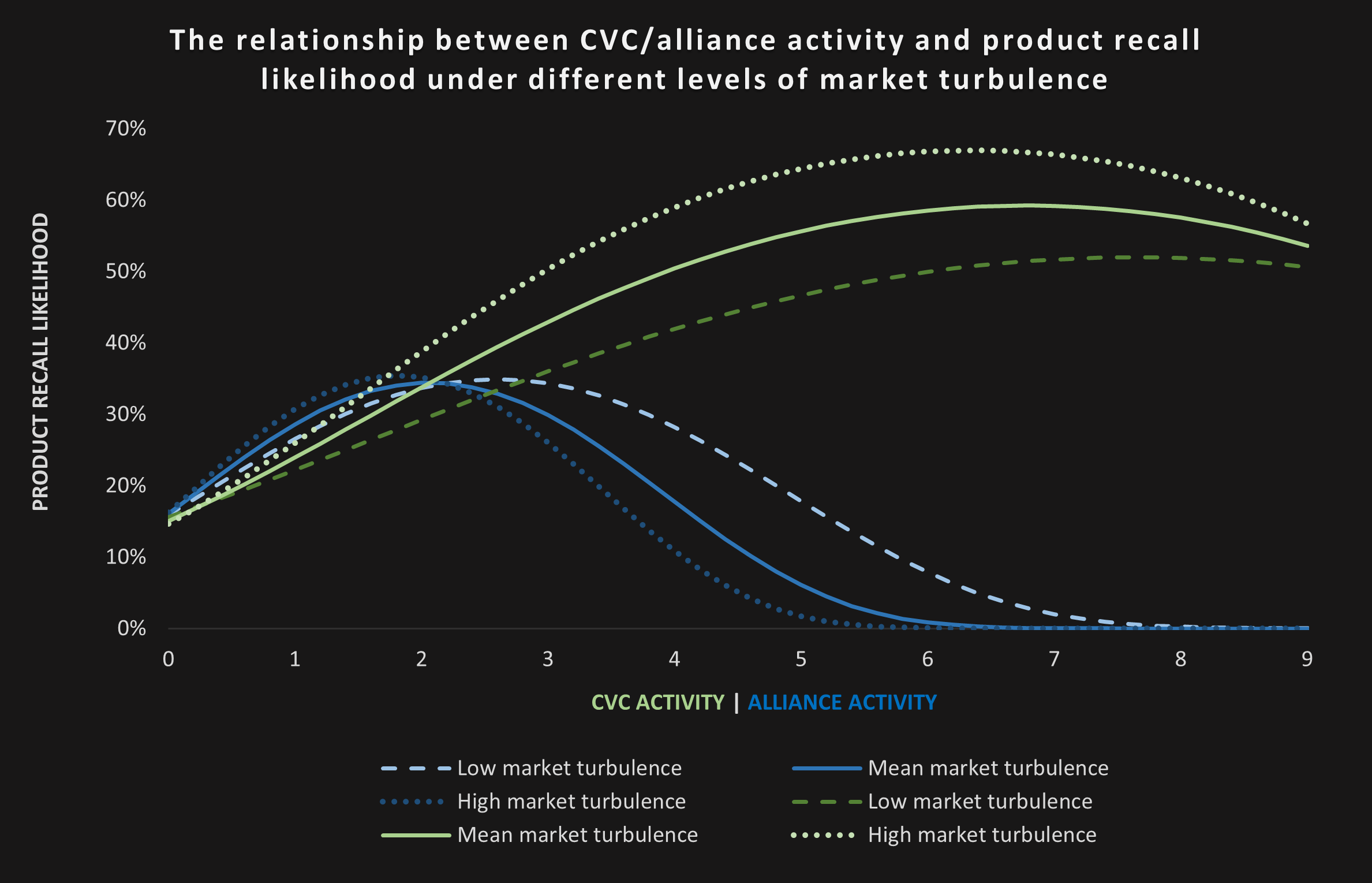
To test our idea, we used longitudinal data from 75 US drug producers and found that the number of firms’ CVC and alliance activities both have an inverted U-shaped relationship with the likelihood of experiencing a product recall in subsequent years. These findings imply that either no or rather bold external venturing strategies pay off more than small-scale “stuck-in-the-middle” initiatives. Hence, firms should either focus on their internal innovation activities or invest boldly into a strong external innovation strategy.
We further deepened our insights by investigating the role of different market environments. We found that high market turbulence makes it even more difficult for firms to benefit from external venturing. While the companies in our sample carried an average standalone recall likelihood of 15-16 percent (without any external venturing), our analyses showed that the recall likelihood increases up to a maximum of 67 percent with six CVC deals and 36 percent with one to two alliances in turbulent markets (versus 52 percent with eight CVC deals and 35 percent between three to four alliances in stable markets). The graph below shows the relationship between CVC and alliance activity with product recall likelihood under different levels of market turbulence.
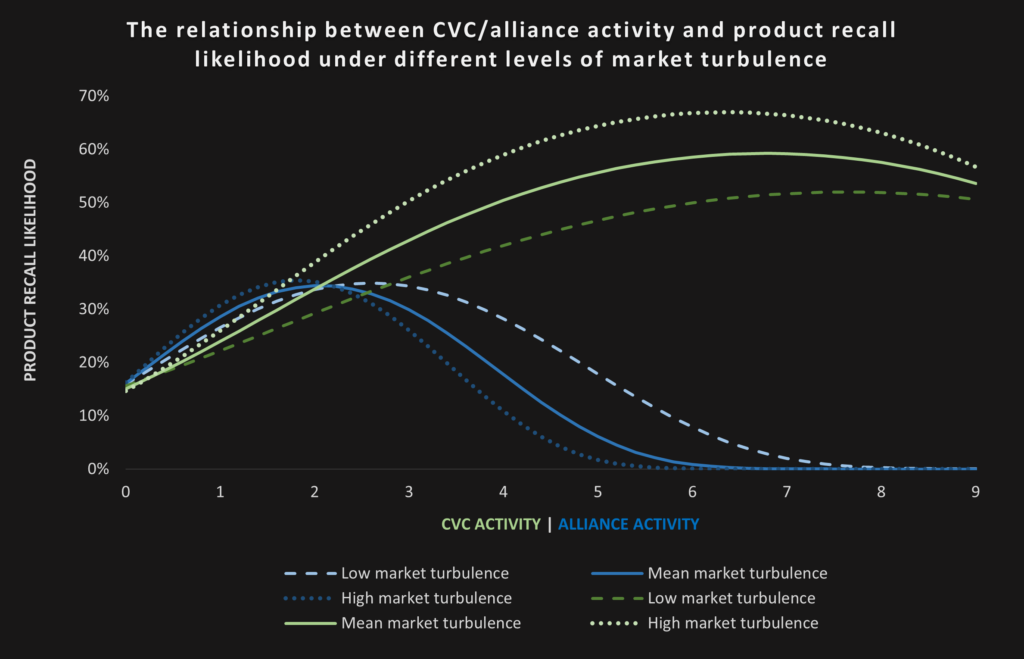
Based on estimates by prior research about the value of CVC deals ($20.4 million) and strategic alliances ($44 to 65 million, as reported by Adegbesan & Higgins, Anand & Khanna, and Kale et al.) as well as the costs of an average recall ($23 million), we also calculated the economic impact of our findings: Firms might lose up to 36 percent of their CVC or 13–19 percent of their alliance deal value through product recalls. Under high market turbulence, this implies recall costs as high as $43.53 million resulting from six CVC deals or $16.74 million from two alliances. Notably, firms with many (six to eight) strategic alliances were able to reduce their recall likelihood to zero under all market conditions, emphasizing the value of a strong alliance strategy for product safety.
Finally, we were able to reproduce our main findings with an independent second sample from the US consumer goods industry, indicating the broader relevance of our findings for different industries as well as policymakers and enforcement agencies such as the Food and Drug Administration or Consumer Product Safety Commission. To identify and remove harmful products promptly, they could monitor firms’ CVC or alliance engagements more closely.